|
The complexes of transition elements of small size are more stable as compared to those of large size elements in a particular series with same oxidation state. This property of formation of complexes is due to vacant d−orbitals (which can accept electron pair) and high nuclear charge with small size which facilitates the acceptance of electrons from the ligands. It must be kept in mind that the ionisation energies of 5d−elements is more than 3d and 4d−elements because of very poor shielding of the nucleus by 4f−electrons.Ĭomplex formation: Transition metal ions have a great tendency to form complexes with other molecules or ions (which can donate a pair of electron to form a coordinate bond) called ligands. Ionisation energy of transition elements in a particular series increases due to increase in nuclear charge. Ionisation Energy: Ionisation energy of transition elements is in between those of The highest possible oxidation state is +8 for transition elements. In lower oxidation states (+2, +3), the bonds formed are ionic, whereas, in higher oxidation states the bonds formed are covalent. The oxidation state of 3rd group elements (Sc, Y, La, Ac) is +3 whereas the oxidation state of 12th group (Zn, Cd, Hg) is +2 only. /metals-including-clockwise-from-left-copper-aluminium-zinc-iron-and-lead-74102855-57ced3d33df78c71b643079f.jpg)
Hence the transition elements exhibit variable oxidation states (except 3rd group and 12th group elements). Oxidation States: During formation of compounds, d−orbitals of inner shell can also participate in bonding. The size of transition elements is small due to which their densities and ionisation energies are high. So their melting and boiling points are very high (more than 1000☌ except Zn, Cd, and Hg), they have high densities and are very hard and brittle.Ītomic Radii: Due to small screening effect of d−subshell the atomic radius decreases with increase in atomic number. They have tendency to form strong covalent bonds due to partially filled d−subshell (except Zn, Cd, Hg) as well as strong metallic bond due to large number of electrons that can be lost. 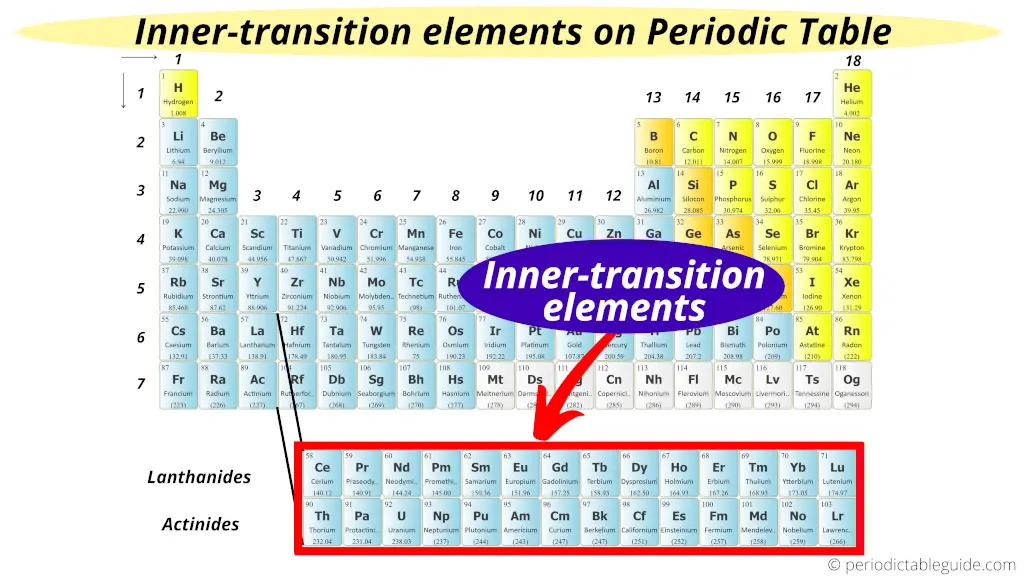
Metallic Character: All the transition elements are metallic in nature. General Characteristics of d−Block Elements The general electronic configuration is On the basis of the principal quantum number of partially filled d−subshell, d−block elements are classified into four series (3d, 4d, 5d and 6d) series, of which 6d series is still incomplete whereas 3d, 4d and 5d series contain 10 elements each and are complete. Transition elements involve the filling of d−subshell of penultimate shell. Further, it must be noted that the elements of group 3 (Sc, Y, La and Ac) differ in their properties from those of other transition elements (they are uniformly trivalent, diamagnetic and colourless). However, they are studied with transition elements just to maintain the status of the periodic table. Moreover, they also do not exhibit general properties of transition elements like variable oxidation states, coloured compounds, formation of complex compounds etc. 
It must be noted that the elements of group 12 (Zn, Cd, and Hg) have completely filled d−subshells in atomic as well as in ionic form, so they do not truly represent transition elements. The transition elements are mainly those elements which either in atomic or ionic state contain partially filled d−subshells. These are called transition elements because they show a gradual gradation in periodic properties. $$ 1s^$ complex ion.In periodic table, the d−block elements are placed between s−block and p−block elements. The electron configuration for titanium is shown below: 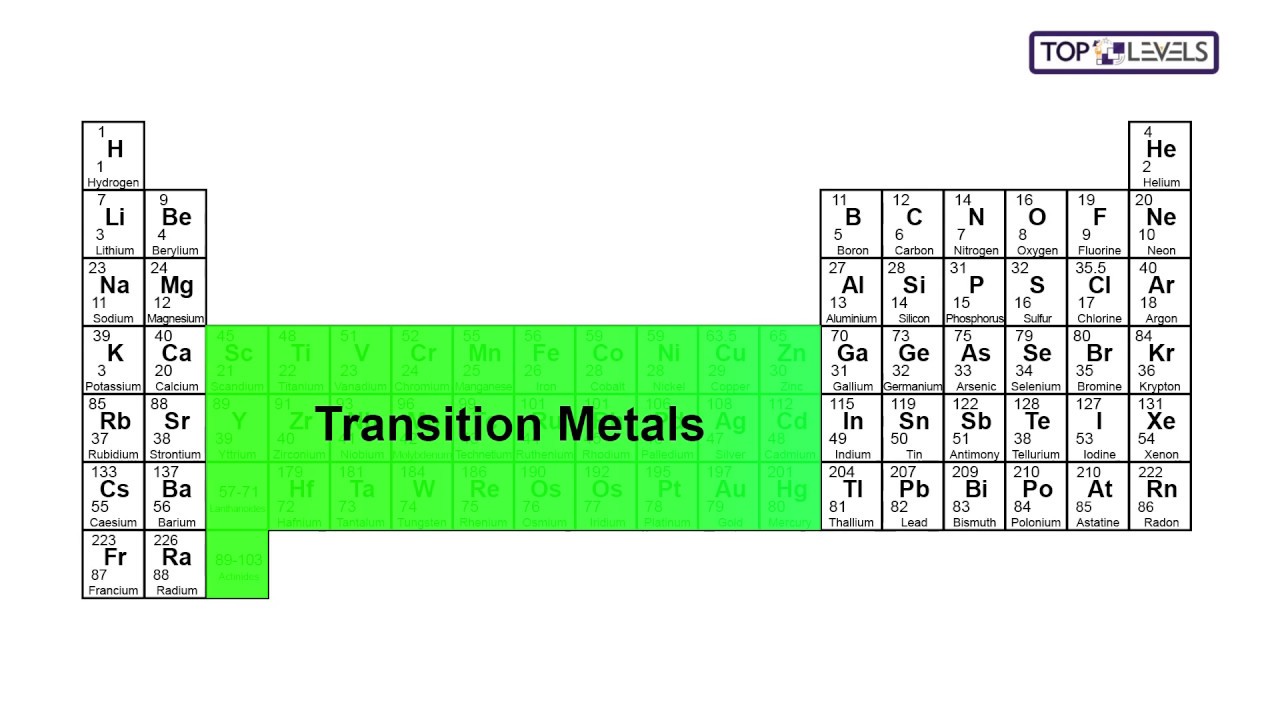
Electron ConfigurationsĮlectron configurations show the arrangement of electrons in an atom of an element. Scandium and zinc are not transition metals as they are unable to generate ions with an incomplete d sub-shell. A transition element is a d-block element that forms at least one ion with an incomplete d sub-shell. The highest electron energy sub-shell in these elements is a d sub-shell. The d-block elements are found in between group 2 and group 3 in the periodic table.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
